A chain of three cell types are involved in converting
light to electrical signals in the optic nerve:
photoreceptors (rods and cones) to bipolar cells to
ganglion cells to cells of the CNS. The purpose of
this retinal arrangement - despite the fact that light
passes through both non-light sensitive and vasculature
structures before photons can be absorbed by
photoreceptors - lies in the link between the outer
segments of photoreceptors and the pigment epithelium.
This thin tissue structure, situated at the back of the
eye with the rod and cone cells functions to remove
photoreceptor disks, because photopigments and proteins
associated with phototransduction have a limited life span
of approximately 12 days. Old outer segment disks are shed
at the periphery of the outer segment, whilst new outer
segment disks are generated along the lowermost portion of
the outer segment. So this means that the molecules of
your eyes associated with vision are shed and renewed
roughly 30 times a year.
Photoreceptors do not demonstrate action potential
patterns that are characteristic of most neurons in the
CNS, but rather have graded changes to membrane potential,
which correlate to changes in neurotransmitter release.
Neuronal signals in the retina only travel short
distances. The absorption of a photon results in the
hyperpolarization of photoreceptors.
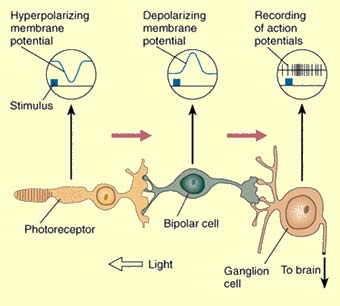

Image credits: Cardiff University
(title); The brain from top to bottom, McGill.
Image credit: Schwartz lab web site, Northwestern
University
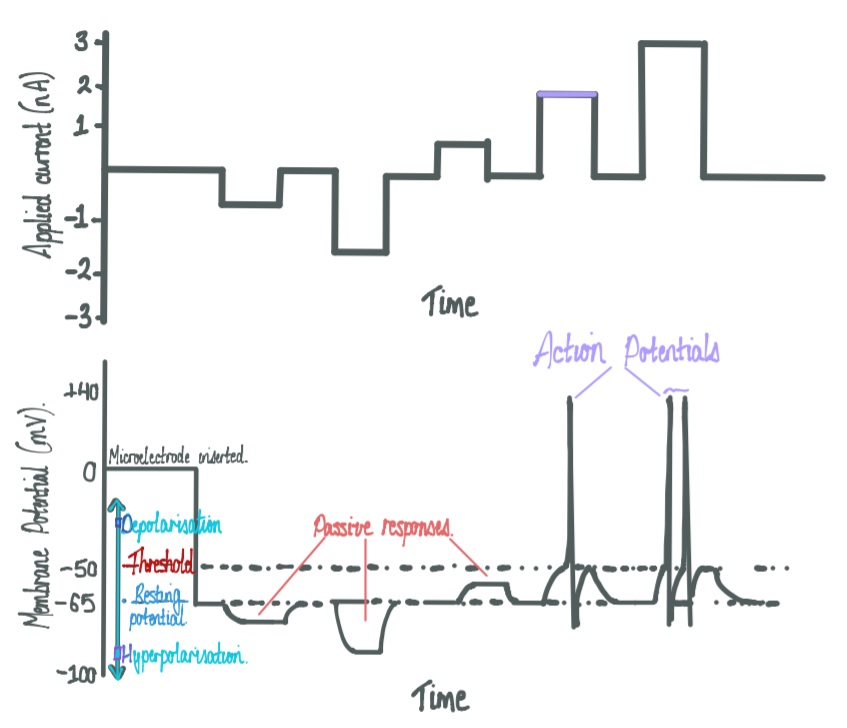
Action potentials are the
specialised electrical signals generated by neurons,
propagated down the length of axons. An action potential is
said to be an
“all-or-none” response to a stimulus –
and is short in duration (~1msec). Upon depolarization of
the membrane, a threshold (mV) must be reached in order to
open sodium gated ion channels, which are
fast-acting, and flood the inside of the cell with sodium
cations. To hyperpolarize the cell, slow-acting potassium
gated ion channel open, and an efflux of potasium cations
returns the cell to its resting membrane potential. When the
membrane is at rest, charge distritution governs ion
permeability, because oppisite charges attract, and because
the movement of ions down their concentration gradient
alters charge distribution. In terms of K+, movement occurs
from the extraceullar to the intraceullar side of the cell
membrane; as more positive charges build-up, net mvoement
stops.
In the absence of an action potential, the
membrane potential is negative (approximately -70 mV) with
respect to the extracellular side. This voltage difference
is set-up due to charge differences associated with
concentration gradients and membrane permeability. The
mechanisms that are the foundation for the resting membrane
potential are: ion concentration gradients; properties of
ion channels and the cell membrane; also, charge
distributions across the membrane. The molecular mechanisms
responsible for the maintenance of the resting membrane
potential include properties of active transport proteins –
namely the Na+/K+ pump, which are membrane-associated
proteins. Eletcrical signals propagate like waves along
threads of axon, connecting to a myrid of connections
through dedritic branches and throughout the CNS via
interneurons. When an electrical signal reaches the end of
an axon, chemical neurtransmitters are releaded into the
synaptic cleft; the chemical reaches the target neurons by
simple diffusion across the synapse. This taregt neuron, in
turn, can spread to another target, and another; the
origianl electrical message can reach billions of neruons
within a few hundred milliseconds, by the time you have
consciously perceived the words on this page.