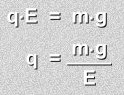The Millikan Oil Drop Experiment
An experiment performed by Robert Millikan in 1909 determined the size of the charge on an electron. He also determined that there was a smallest 'unit' charge, or that charge is 'quantized'. He received the Nobel Prize for his work. We're going to explain that experiment here, and show how Millikan was able to determine the size of a charge on a single electron.
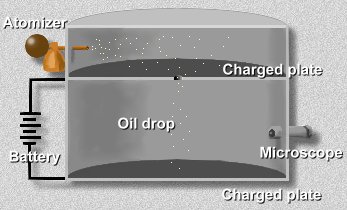
What Millikan did was to put a charge on a tiny drop of oil, and measure how strong an applied electric field had to be in order to stop the oil drop from falling. Since he was able to work out the mass of the oil drop, and he could calculate the force of gravity on one drop, he could then determine the electric charge that the drop must have. By varying the charge on different drops, he noticed that the charge was always a multiple of -1.6 x 10 -19 C, the charge on a single electron. This meant that it was electrons carrying this unit charge.Here's how it worked. Have a look at the apparatus he used:
http://www.mdclearhills.ab.ca/millikan/experiment.html
An atomizer sprayed a fine mist of oil droplets into the chamber. Some of these tiny droplets fell through a hole in the upper floor. Millikan first let them fall until they reached terminal velocity. Using the microscope, he measured their terminal velocity, and by use of a formula, calculated the mass of each oil drop.
Next, Millikan applied a charge to the falling drops by illuminating the bottom chamber with x-rays. This caused the air to become ionized, and electrons to attach themselves to the oil drops.
By attaching a battery to the plates above and below this bottom chamber, he was able to apply an electric voltage. The electric field produced in the bottom chamber by this voltage would act on the charged oil drops; if the voltage was just right, the electromagnetic force would just balance the force of gravity on a drop, and the drop would hang suspended in mid-air.Now you try it. Click here to open a simulation of Millikan's chamber. First, allow the drops to fall. Notice how they accelerate at first, due to gravity. But quickly, air resistance causes them to reach terminal velocity.
Now focus on a single falling drop, and adjust the electric field upwards until the drop remains suspended in mid-air. At that instant, for that drop, the electric force on it exactly equals the force of gravity on it. Some drops have more electrons than others, so will require a higher voltage to stop.
When you've finished playing with the apparatus, close the window and we'll continue.
O.K., let's look at the calculation Millikan was now able to do.When a drop is suspended, its weight m · g is exactly equal to the electric force applied q · E

http://www.mdclearhills.ab.ca/millikan/experiment.html
The values of E, the applied electric field, m the mass of a drop, and g, the acceleration due to gravity, are all known values. So you can solve for q, the charge on the drop:
http://www.mdclearhills.ab.ca/millikan/experiment.html
Millikan determined the charge on a drop. Then he redid the experiment numerous times, each time varying the strength of the x-rays ionizing the air, so that differing numbers of electrons would jump onto the oil molecules each time. He obtained various values for q.
The charge q on a drop was always a multiple of -1.6 x 10 -19 C, the charge on a single electron.This number was the one Millikan was looking for, and it also showed that the value was quantized; the smallest unit of charge was this amount, and it was the charge on a single electron.