When these molecules are heated, different reactions occur and the basic shape of the molecule changes, which is how food is cooked and why it usually changes texture, color, consistency, etc as it is heated.
Proteins
A
protein is a intertwined "chain" of organic
molecules. When heated, these molecules
"unfold", separate, and coagulate. 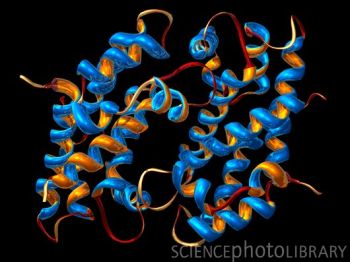
As these molecules unfold, the texture of the food usually firms up. This is why when eggs are overcooked, they
become rubbery. Same goes for how hard and chewy a steak can get if overcooked.
Sugars
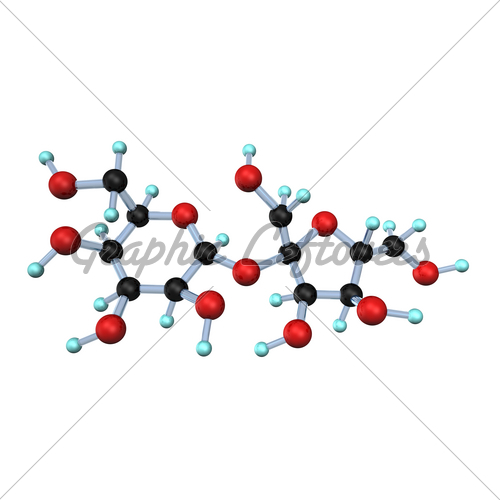 Sugar
molecules are less complex than protein
molecules. When heated, these molecules
undergo a transformation similar
Sugar
molecules are less complex than protein
molecules. When heated, these molecules
undergo a transformation similar
to protein molecules except that they do not necessarily unfold but they do coagulate and release (thus also require) a
lot more energy than protein molecules. This changes the consistency of the food making it "stickier".
The food also gains a browner color. This is because the sugars caramelize as heat is applied.
Fats

As these molecules unfold, the texture of the food usually firms up. This is why when eggs are overcooked, they
become rubbery. Same goes for how hard and chewy a steak can get if overcooked.
Sugars
 Sugar
molecules are less complex than protein
molecules. When heated, these molecules
undergo a transformation similar
Sugar
molecules are less complex than protein
molecules. When heated, these molecules
undergo a transformation similar
to protein molecules except that they do not necessarily unfold but they do coagulate and release (thus also require) a
lot more energy than protein molecules. This changes the consistency of the food making it "stickier".
The food also gains a browner color. This is because the sugars caramelize as heat is applied.
Fats
These
molecules are chains of organic molecules
that do not mix with water and therefore
do not evaporate. Fats melt when heated.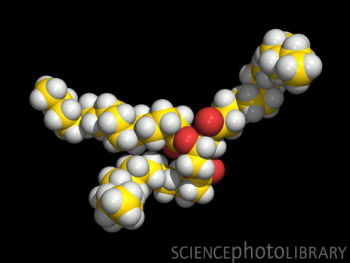
Water
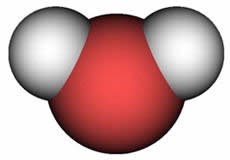 All foods
contain water. Water molecules evaporate
when heated to a certain temperature.
This dries out the food
All foods
contain water. Water molecules evaporate
when heated to a certain temperature.
This dries out the food
and creates air pockets where the water molecules used to be. This is why you see steam when cooking foods, especially on
the stove top. The evaporated water molecules are escaping from the food.
Starches
When starch and liquid is heated, starch granules expand and thicken. This thickens the overall consistency of the starch and liquid
mixture because the expanded starch molecules occupy more and more space. This is why bread rises (and has air pockets)
as it bakes. The water molecules evaporate while starch molecules expand.
(Photo credits: janielsantos3b.edublogs.org and sciencephotz

Water
 All foods
contain water. Water molecules evaporate
when heated to a certain temperature.
This dries out the food
All foods
contain water. Water molecules evaporate
when heated to a certain temperature.
This dries out the food and creates air pockets where the water molecules used to be. This is why you see steam when cooking foods, especially on
the stove top. The evaporated water molecules are escaping from the food.
Starches
When starch and liquid is heated, starch granules expand and thicken. This thickens the overall consistency of the starch and liquid

mixture because the expanded starch molecules occupy more and more space. This is why bread rises (and has air pockets)
as it bakes. The water molecules evaporate while starch molecules expand.
(Photo credits: janielsantos3b.edublogs.org and sciencephotz